The prognostic significance of combined ERG and androgen receptor expression in patients with prostate cancer managed by androgen deprivation therapy
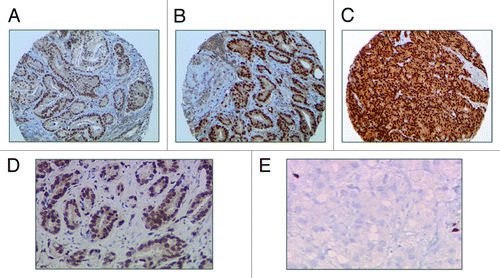
Cancer Biol Ther. 2014 Sep;15(9):1120-8. doi: 10.4161/cbt.29689. Epub 2014 Jun 27.
Huang KC1, Alshalalfa M2, Hegazy SA3, Dolph M1, Donnelly B4, Bismar TA5.
Abstract
ERG and androgen receptor (AR) are known to function cooperatively in prostate cancer (PCa) progression. However, the prognostic value of combined ERG and AR expression and potential pathways are not well characterized. We assessed ERG and AR protein expression by immunohistochemistry in a cohort of 312 men with PCa diagnosed by transurethral resection of the prostate (TURP). Patients were divided into those with no prior hormonal treatment (designated as PCa/AdvPCa) vs. those with castrate-resistant PCa (CRPC) undergoing channel TURP to relieve obstructive symptoms. The expression status was correlated with various clinical-pathological parameters. The Swedish watchful-waiting cohort was used for validation and characterization of potential gene signatures associated with ERG and AR. Patients with combined ERG-positive/AR high expression profile demonstrated higher rates of PCa-specific mortality (PCSM) compared with patients with ERG-negative/AR low in patients with no prior treatment (n = 90, P = 0.032), but this was attenuated in the overall cohort which included the CRPC subgroup (n = 125, P = 0.096). The prognostic significance to PCSM was validated in the Swedish watchful waiting cohort in univariate (HR: 3.3; 95% CI: 1.9-5.6, P = 4.25E-5) and multivariate analysis (HR: 2; 95% CI: 0.97-4.1, P = 0.057), which included Gleason score. ERG/AR overexpression status characterized 152 genes signatures including WNT, PI3K/AKT and chemokine signaling pathways known to be deregulated in PCa. In conclusion, combined ERG/AR overexpression signifies a class of patients at highest-risk of PCSM with specific key genetic alteration likely responsible for disease progression. The prognostic value of combined ERG/AR overexpression and its associated genes should be further investigated as potential prognostic and therapeutic targets in prostate cancer progression.
